Ketone and Aldehyde α-Oxygenation
The α-hydroxycarbonyl moiety is an important structural motif in organic synthesis and is present in a substantial number of natural products. A variety of methods have been developed to prepare this functionality including α-oxygenation of enolates with electrophilic oxidizing agents, as well as dihydroxylation or epoxidation of enol ethers.1 Recently, aminooxylation of aldehydes2 and ketones3 has been demonstrated using nitrosobenzene. However, this latter protocol requires a large excess of the carbonyl compound in tandem with syringe pump techniques that diminishes the usefulness of the transformation.
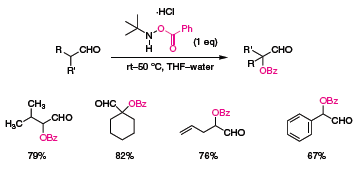
Nicholas Tomkinson of Cardiff University (UK) recently reported several practical reagents for the preparation of α-oxygenated carbonyl compounds.1 N-tert-butyl-O-benzoylhydroxylamine hydrochloride is a bench stable reagent for the α-functionalization of a variety of aldehydes under mild conditions (Scheme 1). The reaction can be performed in open air and in the presence of moisture. Benzoylation occurs in high-yield at both secondary and tertiary centers as demonstrated by the reactions of this reagent with isovaleraldehyde and cyclohexanecarboxaldehyde.
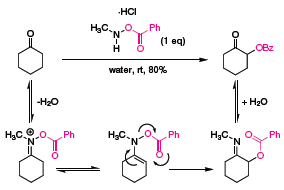
While this reagent is only useful on aldehyde substrates, a similar bench stable reagent can be applied towards the α-oxygenation of ketones. Treatment of cyclohexanone with N-methyl-O-benzoylhydroxylamine hydrochloride in water provides the benzoyl-protected alcohol in excellent yield, presumably through a pericyclic rearrangement of an intermediate iminium ion (Scheme 2). A previous synthesis of this molecule by House required a laborious five-step procedure.5
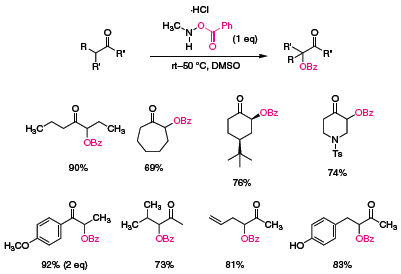
Both acyclic and cyclic ketones undergo oxidation at mild temperatures to afford protected alcohols in good to excellent yield (Scheme 3). The reagent is tolerant of existing functionality (e.g. sulfonamides, free hydroxyl groups, and unsaturation), and in the case of unsymmetrical ketones, oxidation occurs regiospecifically at the more substituted carbon. In addition to water and DMSO, THF and CHCl3 can be used as solvents with similar yields.
We are delighted to add these new tools to our ever-growing arsenal of oxidation reagents.
Materials
References
To continue reading please sign in or create an account.
Don't Have An Account?